RenalGLO™
An In Vitro Bioluminescence Assay for Renal Viability and Function
Buy Add-On Standardization Assay Kit
| Description | Catalog Number | Quantity |
|---|---|---|
| An Add-On Kit for Any Bioluminescence "GLO" Assay | K-ATPSC-1 | 1 Kit |
RenalGlo™
The Easy and Rapid In Vitro Research Bioluminescence Assay for Kidney Cells
| Cell Population | Species | Medium Formulation | Catalog Number | Quantity |
|---|---|---|---|---|
| Renal cells (adherent) | Any | User defined | KRG-A-1 | 1 Kit |
| Renal cells (non-adherent) | Any | User defined | KRG-NA-1 | 1 Kit |
Uses of RenalGlo™
- Determine the metabolic viability of different kidney cell types.
- Study factors produced by different kidney cells types in culture, e.g. erythropoietin.
- Determine metabolic/mitochondrial dysfunction in kidney cells.
- Study the effect of agents on kidney function in different renal cell types.
- Study mechanisms of renal injury.
- Study renal disorders at the cellular and genetic levels.
- The most sensitive and accurate assay readout available incorporating proven ATP bioluminescence technology.
- Optional assay standardization using the ATP Bioluminescence Standardization & Calibration Kit (K-ATPSC-1).
- Measurement assurance parameters ensure the assay is working correctly.
- Validated according to FDA bioanalytical method validation guidelines.
- Fast, reliable and reproducible readout with result in 15 minutes for a 96-well plate
- High throughput capability.
- Reliably compare results over time.
- Multiplexing capability allowing the assay to be combined with other endpoints from the same sample.
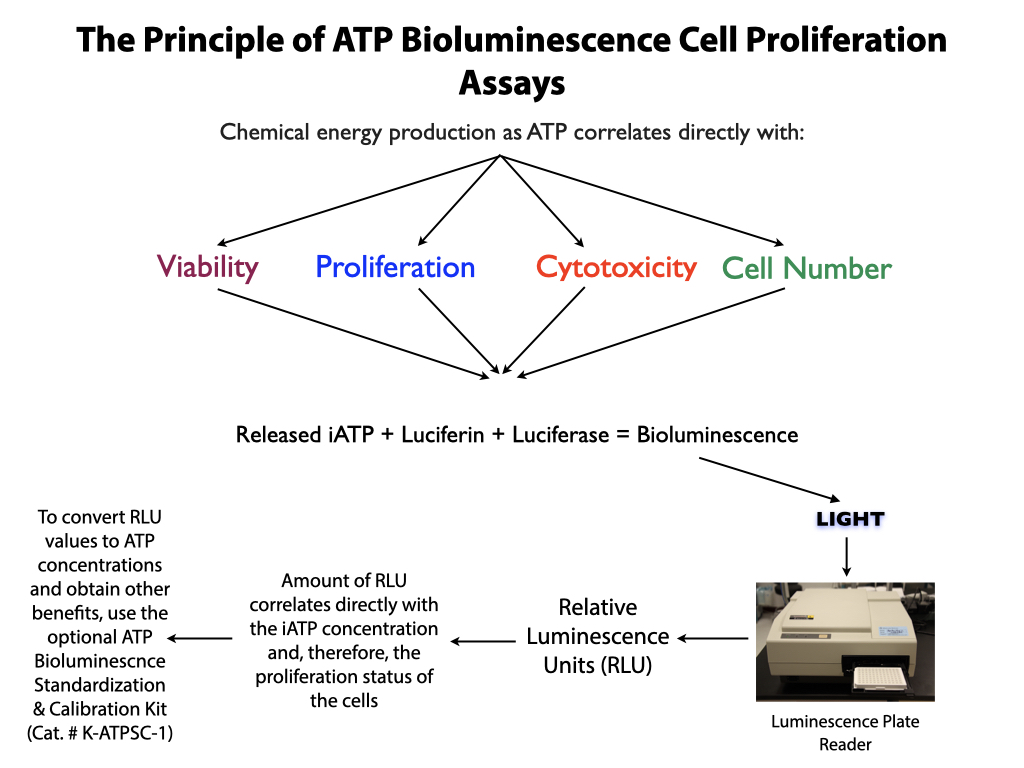
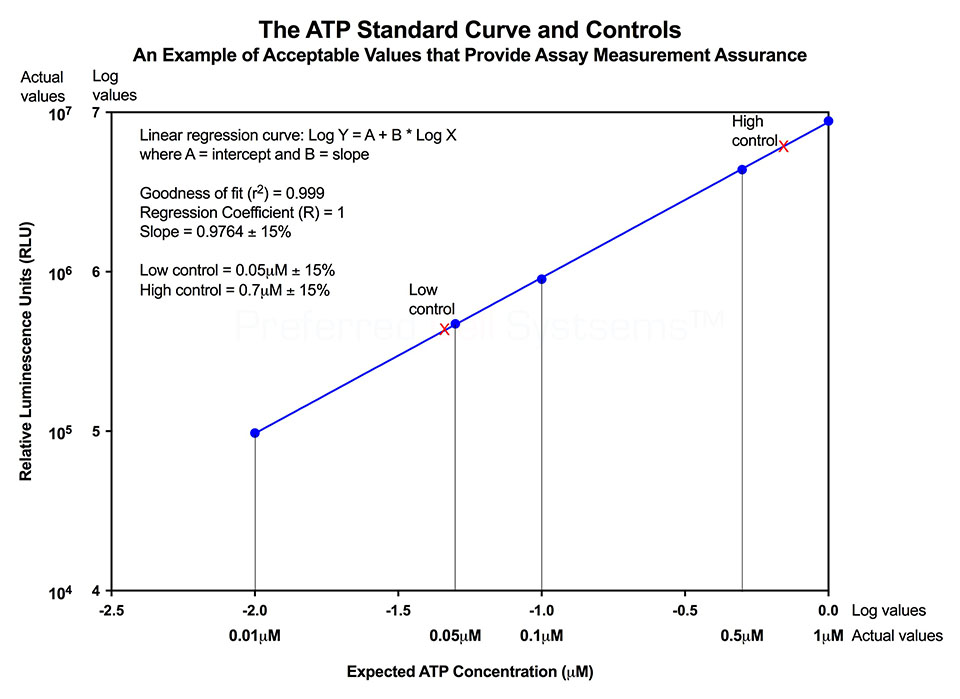
Performing the ATP standard curve and controls is highly recommended, since non-standardized RLU values can be converted into standardized ATP concentrations. The controls are used to calibrate the luminescence plate reader and the ATP standard is used to standardize the assay. The values obtained from the standard curve and controls (see diagram below) are then compared to the measurement assurance parameters that are provided in the instruction manual. If the values obtained are within the ranges provided by the measurement assurance parameters, you, the user, have successfully performed the proficiency testing to ensure that you have not only performed the assay correctly, but that the results you obtain can be trusted.
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.
Species and Cell Sources
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.

RenalGlo™ can be used for multiple species. Primary kidney cells as well as kidney cell lines, such as HepG2. Many kidney cell types have a high level of metabolic activity that can be used to determine changes in metabolic viability and other functions.
Major Equipment Needed
For Research Use Only. Not for clinical diagnostic use.
Luminescence or multimode plate reader with "glow" luminescence measuring capability.
- ATP Enumeration Reagent.
- Sterile, white, 96-well plates for kidney culture. Choice of adherent or non-adherent plates.
- Sterile, adhesive foil covers to maintain sterility of unused wells.
PLEASE NOTE. The investigator has the flexibility to culture renal cells using their own growth medium. RenalGro™ Growth Medium is available separately from Preferred Cell Systems™.
Please visit the Instructional Videos page
Download the RenalGlo™ Technical Manual
Download the ATP Bioluminescence Standardization & Calibration Kit Manual
Download Certificate of Analysis (CoAs) for ATP Enumeration Reagent (ATP-ER)
Download Certificate of Analysis for ATP Stanadrds
Download Certificate of Analysis for ATP Controls
Download Certificate of Analysis of ATP Reconstitution Reagent
Download Certificate of Analysis for Sterile 96-Well Plates
Download Certificate of Analysis for Non-Sterile, 96-Well Plates
Download Certificate of Analysis for Sealing Films
Download Certificate of Analysis for IMDM
Download the RenalGlo™ Technical Manual
Download the ATP Bioluminescence Standardization & Calibration Kit Manual
Download Certificate of Analysis (CoAs) for ATP Enumeration Reagent (ATP-ER)
Download Certificate of Analysis for ATP Stanadrds
Download Certificate of Analysis for ATP Controls
Download Certificate of Analysis of ATP Reconstitution Reagent
Download Certificate of Analysis for Sterile 96-Well Plates
Download Certificate of Analysis for Non-Sterile, 96-Well Plates
Download Certificate of Analysis for Sealing Films
Download Certificate of Analysis for IMDM
