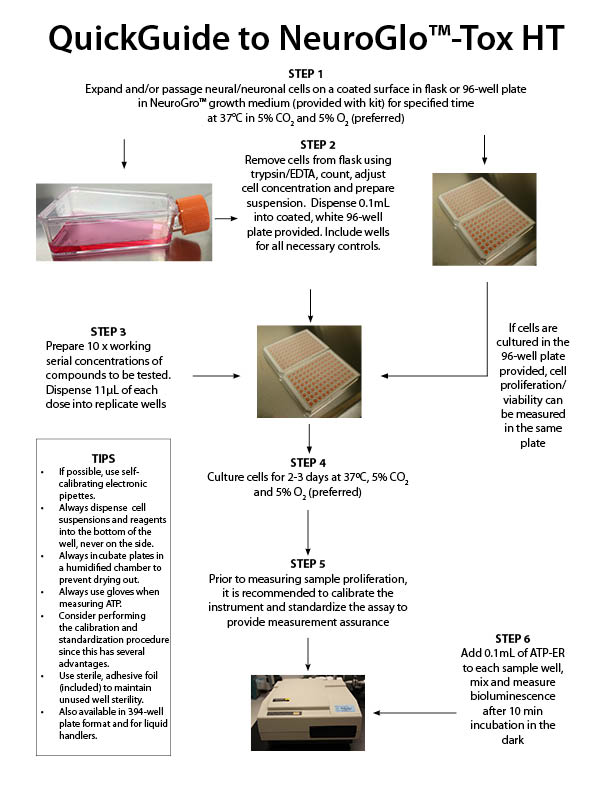
NeuroGlo™-Tox HT
An In Vitro Standardized and Validated
ATP Bioluminescence Neurotoxicity
Testing and High-Throughput Screening Platform
Buy NeuroGLO™-Tox HT
NeuroGLO™-Tox HT
A Standardized and Validated In Vitro Bioluminescence Neurotoxicity Assay
| Cell Population | Species | Medium Formulation | Catalog Number | Quantity |
|---|---|---|---|---|
| Neural cells (adherent) | Any | User defined | KNG-T96A-4 | 4 Plates/1Kit |
| Neural cells (non-adherent) | Any | User defined | KNG-T96NA-4 | 4 Plates/1Kit |

- Viability, cell functionality and cell number assays for neural cells.
- Developmental biology.
- Determine expansion ability and potential.
- Neural proliferation and differentiation process.
- Comparison of ES-derived cells with other neural or neuronal cells.
- High throughput in vitro toxicity testing and screening.
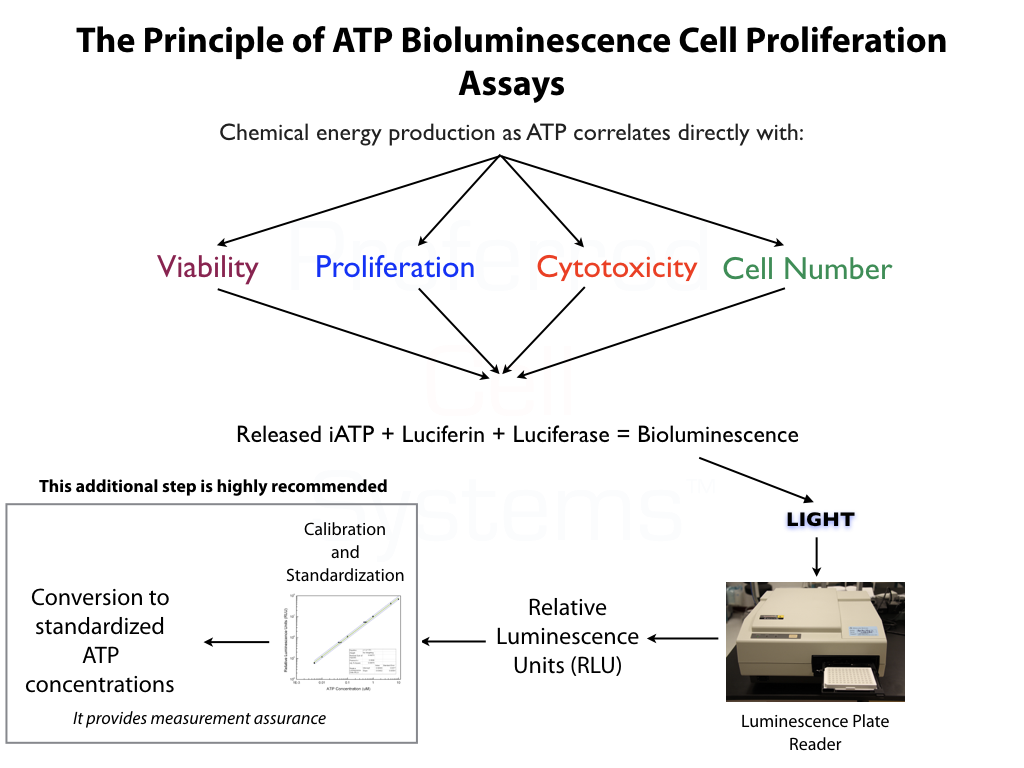
- Incorporates the most sensitive of any assay readout using proven ATP-based bioluminscence technology.
- Instrument-based, quantitative and fully standardized readout procedure that can be validated according to regulatory requirements.
- High throughput capability.
- Reliably compare results over time.
- Multiplexing capability to incorporate other assay readouts for additional information from the same sample.
- Simple, time efficient and cost effective.
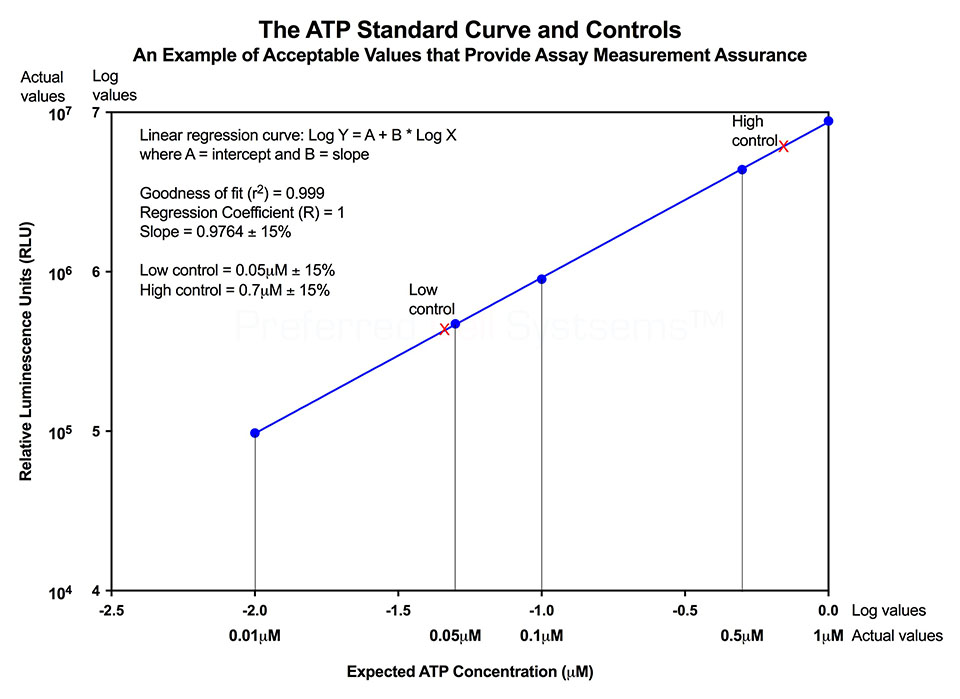
All ATP bioluminescence assays kits from Preferred Cell Systems™ include an ATP standard(s) and controls. Performing the ATP standard curve and controls is highly recommended. The controls are used to calibrate the luminescence plate reader and the ATP standard is used to standardize the assay. The values obtained from the standard curve and controls (see diagram below) are then compared to the measurement assurance parameters that are provided in the instruction manual. If the values obtained are within the ranges provided by the measurement assurance parameters, you, the user, have successfully performed the proficiency testing to ensure that you have not only performed the assay correctly, but that the results you obtain can be trusted.
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.
Species and Cell Sources
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.

- Virtually any mammalian species.
- Primary or cryopreserved.
For Research Use Only. Not for clinical diagnostic use.
Luminescence or multimode plate reader with "glow" luminescence measuring capability.
- Medium for dilution of ATP standard
- ATP standard
- ATP controls
- ATP Enumeration Reagent
- Sterile, 96-plates. (384-well plates are available upon request)
- Non-sterile, 96-plate for standard curve and controls. (384-well plates are available upon request).
- Sterile, adhesive foil covers to maintain sterility of unused wells
Please visit the Instructional Videos page
Download the NeuroGlo™-Tox HT Technical Manual
Download the ATP Optimization Kit Protocol for First-Time Users
Download Luminometer Setup and RLU to ATP Conversion
Download Certificate of Analysis (CoAs) for ATP Enumeration Reagent (ATP-ER)
Download Certificate of Analysis for ATP Stanadrds
Download Certificate of Analysis for ATP Controls
Download Certificate of Analysis of ATP Reconstitution Reagent
Download Certificate of Analysis for Sterile 96-Well Plates
Download Certificate of Analysis for Non-Sterile, 96-Well Plates
Download Certificate of Analysis for Sealing Films
Download Certificate of Analysis for IMDM
Download the NeuroGlo™-Tox HT Technical Manual
Download the ATP Optimization Kit Protocol for First-Time Users
Download Luminometer Setup and RLU to ATP Conversion
Download Certificate of Analysis (CoAs) for ATP Enumeration Reagent (ATP-ER)
Download Certificate of Analysis for ATP Stanadrds
Download Certificate of Analysis for ATP Controls
Download Certificate of Analysis of ATP Reconstitution Reagent
Download Certificate of Analysis for Sterile 96-Well Plates
Download Certificate of Analysis for Non-Sterile, 96-Well Plates
Download Certificate of Analysis for Sealing Films
Download Certificate of Analysis for IMDM