STEMGlo™-Tox HT
Buy STEMGlo™-Tox HTSTEMGlo™-Tox HT
A Standardized and Validated In Vitro Bioluminescence Toxicity Assay for Stem Cell Lines
| Cell Population | Species | Medium Formulation | Catalog Number | Quantity |
|---|---|---|---|---|
| ES, iPS and other stem cell lines (adherent) | Any | User defined | KSG-T96A-4 | 1 Kit |
| ES, iPS and other stem cell lines (non-adherent) | Any | User defined | KSG-T96NA-4 | 1 Kit |
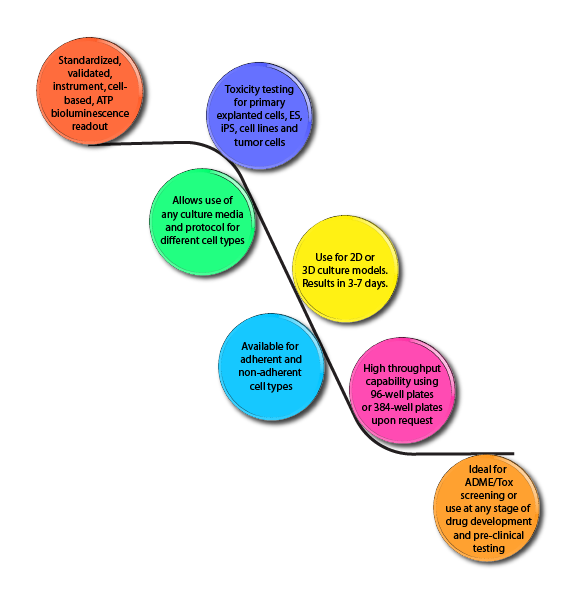
Fully Standardized ATP Bioluminescence In Vitro Toxicity Assay Platform for ES, iPS or other stem cell lines.
Uses
- High throughput in vitro toxicity screening and testing using embryonic stem (ES), induced pluripotent stem (iPS) cells or other stem cell lines.
- Can be used for most transformed cell lines.
- Can be incorporated at any stage of drug development, including ADME/Tox.
- Drug/agent toxicity ranking according to cell source and species.
- Development of drugs and other therapies using stem cell gene targeting and expression research, e.g. CRISPR-Cas9.
- A flexible, in vitro assay platform to perform high throughput toxicity testing.
- Can be applied to numerous cell types derived from different organs, tissues and species.
- High-throughput capability using 96- or 384-well plate formats allows ADME-Tox drug or compound screening, thereby significantly reducing unexpected results during pre-clinical testing.
- A 3Rs Assay Platform-Helps Reduction, Refinement, Replacement for animal testing.
- Available for adherent or non-adherent cell types.
- 2D and 3D cell culture models.
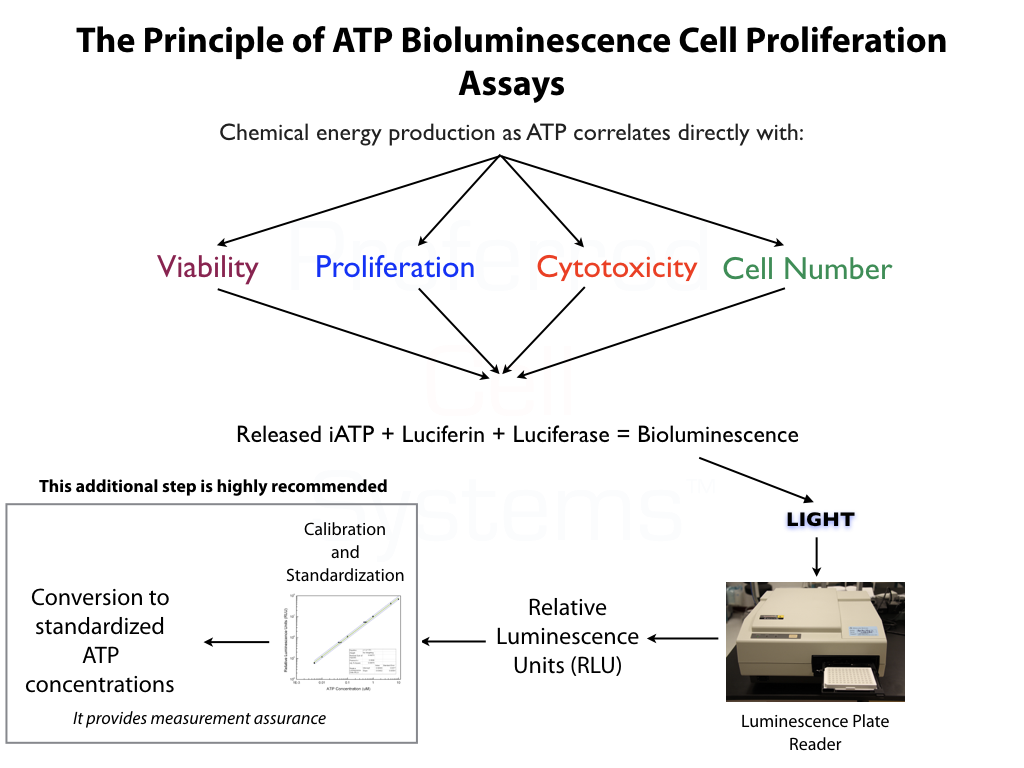
- Incorporates the most sensitive ATP bioluminescence readout available to measure proliferation, cytotoxicity, cell number and even apoptosis.
- Kits include standards and controls to calibrate and standardize the assay.
- Validated signal detection readout according to FDA Bioanalytical Method Guidelines.
- Allows results to be compared over time.
- Results usually in 4 to 5 days depending on species and cell type.
- Designed for multiplexing with other assay using the same sample.
- Simple, time efficient and cost effective.
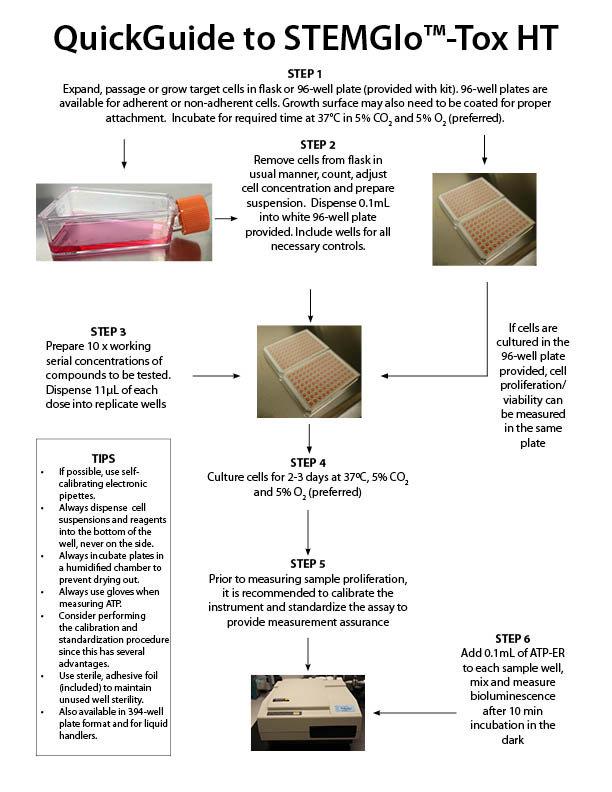
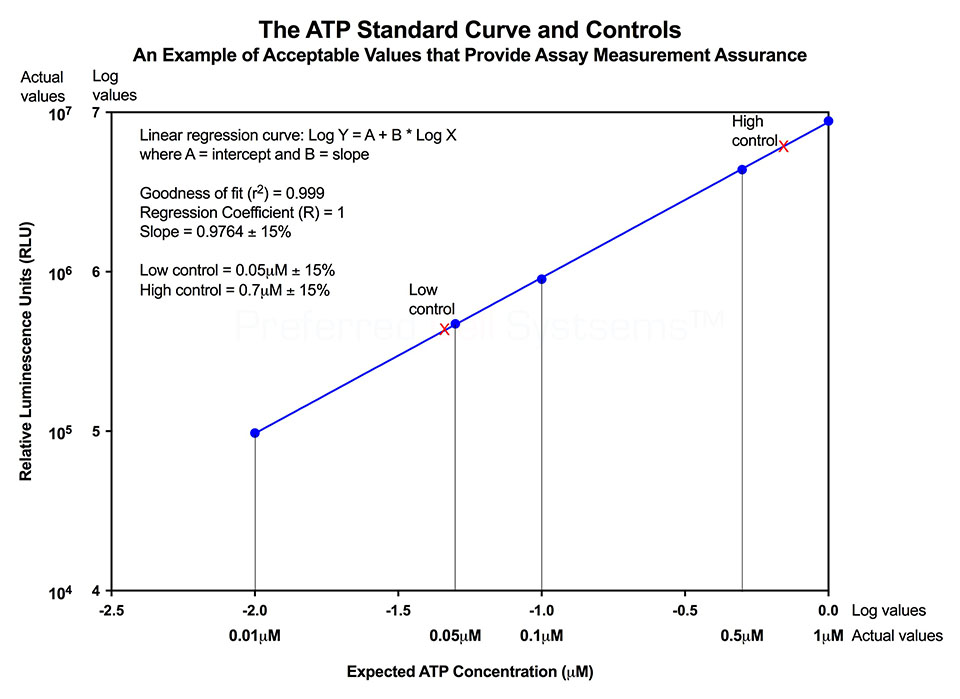
All ATP bioluminescence assays kits from Preferred Cell Systems™ include an ATP standard(s) and controls. Performing the ATP standard curve and controls is highly recommended. The controls are used to calibrate the luminescence plate reader and the ATP standard is used to standardize the assay. The values obtained from the standard curve and controls (see diagram below) are then compared to the measurement assurance parameters that are provided in the instruction manual. If the values obtained are within the ranges provided by the measurement assurance parameters, you, the user, have successfully performed the proficiency testing to ensure that you have not only performed the assay correctly, but that the results you obtain can be trusted.
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.
Species and Cell Sources
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.

STEMGlo™-Tox HT can be used to investigate potential in vitro toxicity using ES, iPS or other stem cell lines for different species as well as virtually any transformed cell lines from any source or species.
Major Equipment Needed
For Research Use Only. Not for clinical diagnostic use.
Luminescence or multimode plate reader with "glow" luminescence measuring capability.
- ATP standard
- ATP controls
- Medium
- ATP Enumeration Reagent
- Sterile, white solid, 96-well plates. Available for adherent or non-adherent cells.
- Non-sterile, 96-well plates for assay calibration and standardization.
- Sterile, adhesive foil covers to maintain sterility of unused wells