EpiCLONE™
Buy EpiClone™EpiClone™
A Standardized and Validated Methylcellulose Clonal Bioluminescence Assay for Intestinal Cells
| Cell Population | Species | Medium Formulation | Catalog Number | Quantity |
|---|---|---|---|---|
| Intestinal cells (adherent) | Any | User defined | KEC-A-1 | 1 Kit |
| Intestinal cells (non-adherent) | Any | User defined | KEC-NA-1 | 1 Kit |
An In Vitro Methylcellulose Clonal and
Bioluminescence Assay for Intestinal Cells
Uses of EpiCLONE™
- To determine cell clonogenicity (colony-forming ability) of intestinal stem and progenitor cells.
- To determine the proliferation and differentiation ability and/or potential of intestinal stem or progenitor cells.
- Allows correlation between proliferation and differentiation ability.
- Clonal (colony-forming) assay standardization.
- Identify factors that affect intestinal stem cells.
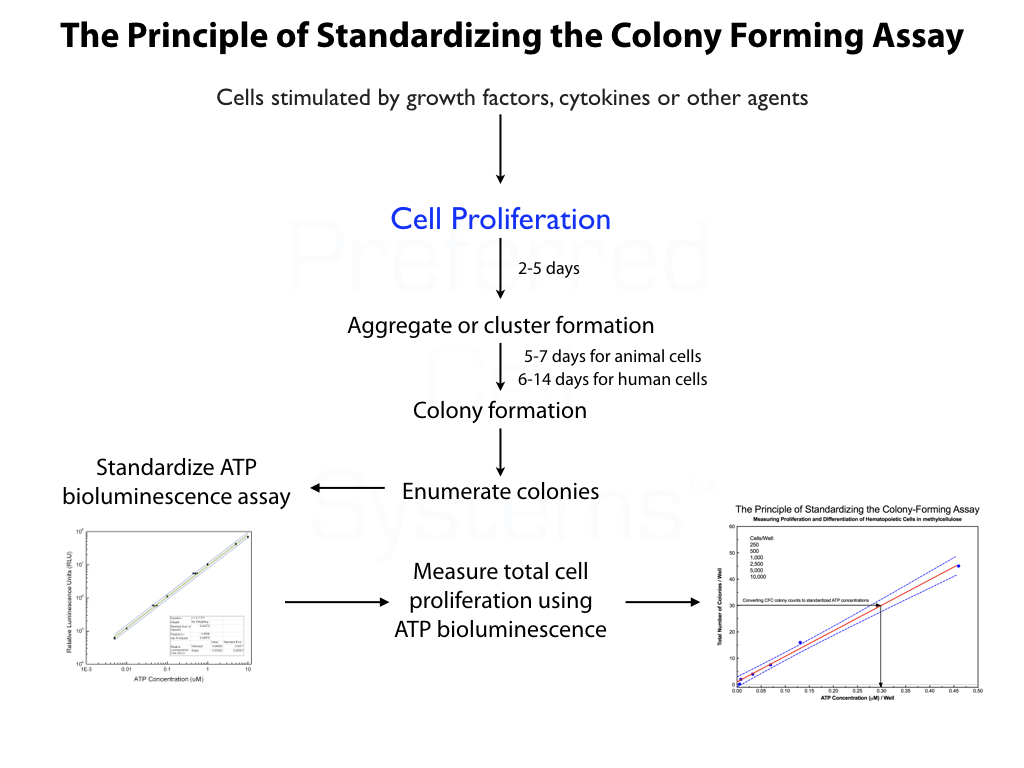
- Duel readouts in a single assay. Cells are allowed to form colonies which can be counted. Proliferation within the colonies can be quantified using an ATP bioluminescence readout
- The ATP bioluminescence readout measures cell proliferation which can be standardized to allow results to be compared from different experiments.
- Due to the correlation between colony counts (differentiation) and ATP concentration (cell proliferation), the assay allows subjective colony counts to be expressed as standardized ATP concentrations.
- Reduces subjective colony counting high variability to a quantitative assay with low variability.
- Incorporates proven standardized ATP bioluminescence technology used in many assays from Preferred Cell Systems™.
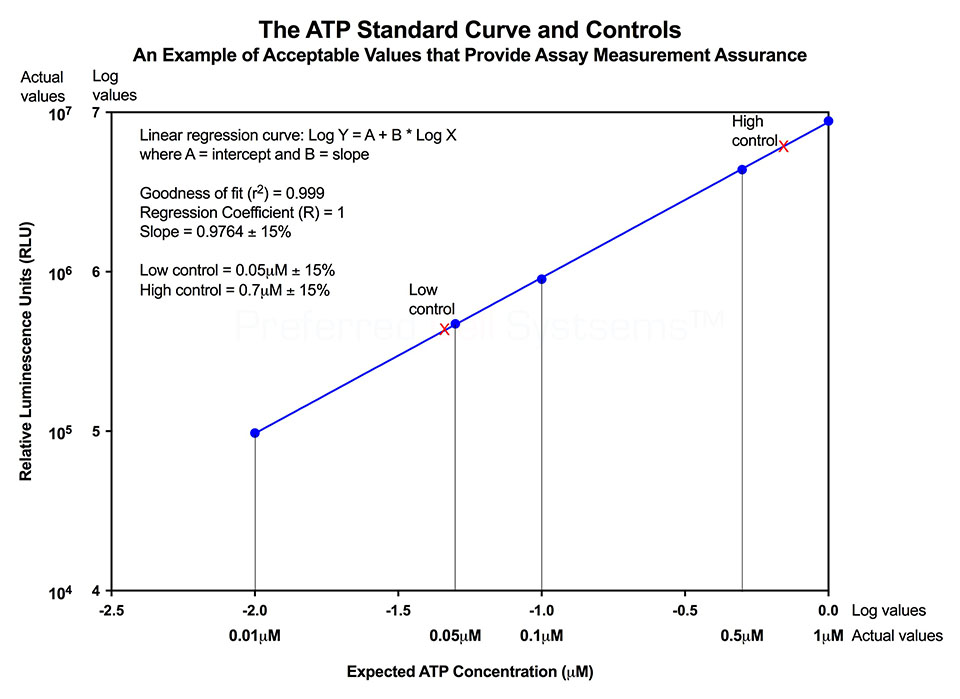
All ATP bioluminescence assays kits from Preferred Cell Systems™ include an ATP standard(s) and controls. Performing the ATP standard curve and controls is highly recommended. The controls are used to calibrate the instrument and the ATP standard is used to standardize the assay. The values obtained provide the measurement assurance parameters that allow you, the user, to ensure that the results obtained can be trusted.
An example of an ATP standard curve and control results are shown in the diagram below and actual values for the kit being used are given in the Technical Manual. If the values obtained are within the ranges given in the Technical Manual, the user can process the samples knowing that the results will be trustworthy.
Species and Cell Sources
An example of an ATP standard curve and control results are shown in the diagram below and actual values for the kit being used are given in the Technical Manual. If the values obtained are within the ranges given in the Technical Manual, the user can process the samples knowing that the results will be trustworthy.

EpiCLONE™ can be used for clonality of intestinal stem and progenitor cells from multiple species.
Major Equipment Needed
For Research Use Only. Not for clinical diagnostic use.
An inverted microscope with a 10x objective and at least a 4x - 10x occular lens is required.
Luminescence or multimode plate reader with "glow" luminescence measuring capability.
- EpiCLONE™ Methylcellulose Base Reagent.
- ATP standard.
- ATP controls.
- ATP Enumeration Reagent.
- Sterile, white, clear-bottom 96-well plates. Available with a non-adherent or adherent growth surface.
- Non-sterile, 96-well plates.
- Sterile, adhesive foil covers to maintain sterility of unused wells.