HemoFLUOR™ PCA
The Fluorescence Progenitor Cell Assay (PCA)
that Replaces Methylcellulose CFU for the
Cell Processing Laboratory
Buy HemoFLUOR™ PCA
HemoFLUOR™ PCA
An Easy and Rapid Methylcellulose-Free Absorbance Progenitor Cell Assay for the Cell Processing Laboratory
| Cell Population | MethoCult Equivalent | Formulation | Catalog Number | Quantity | |
|---|---|---|---|---|---|
| Stem Cells | |||||
| SC-GEMM 3 | H4435 "Enriched" | Low serum | K5-PCA5-1 | 1 Kit | |
| SC-GEMM | MethoCult "Express" | Low serum | K5-PCA6-1 | 1 Kit | |
| SC-GEM 3 | H4434 "Classic" | Low serum | K5-PCA1-1 | 1 Kit | |
| SC-GEM 2 | H4034 "Optimal" | Low serum | K5-PCA2-1 | 1 Kit | |
| Progenitor Cells | |||||
| P-GM 1 | H4534 "Classic" | Low serum | K5-PCA3-1 | 1 Kit | |
| P-GM 2 | H4035 "Optimal" | Low serum | K5-PCA4-1 | 1 Kit | Stem Cells |
| SC-GEMM 3 | H4435 "Enriched" | Serum-Free | K5SF-PCA5-1 | 1 Kit | SC-GEMM | Metocult "Express" | Serum-Free | K5SF-PCA6-1 | 1 Kit | SC-GEM 3 | H4434 "Classic" | Serum-Free | K5SF-PCA1-1 | 1 Kit | SC-GEM 2 | H4034 "Optimal" | Serum-Free | K5SF-PCA2-1 | 1 Kit |
| Progenitor Cells | |||||
| P-GM 1 | H4534 "Classic" | Serum-Free | K5SF-PCA3-1 | 1 Kit | |
| P-GM 2 | H4035 "Optimal" | Serum-Free | K5SF-PCA4-1 | 1 Kit | |

Instructional Video on How to Use HemoFLUOR™ PCA
To learn how to setup and use HemoFLUOR™ PCA, click on the video links below:
How to Perform a Progenitor Cell Assay (PCA) - An Alternative to All Methylcellulose CFU Assays

- A fluorescence, CFU-equivalent assay for cell processing laboratories.
- No colony counting required.
- Transforms the subjective CFU assay into an instrument-based, quantitative assay.
- Incorporates the same growth factor cocktails used in MethoCult® reagents for direct assay-to-assay comparisons and correlation.
- Non-subjective, instrument-based and quantitative.
- Results in 7 days.
- Faster, easier and more cost effective to use than any traditional CFU assay, even using automated colony counting.
- Complete assay kit; includes everything needed to culture and obtain results.
- Permitted by FDA and AABB.
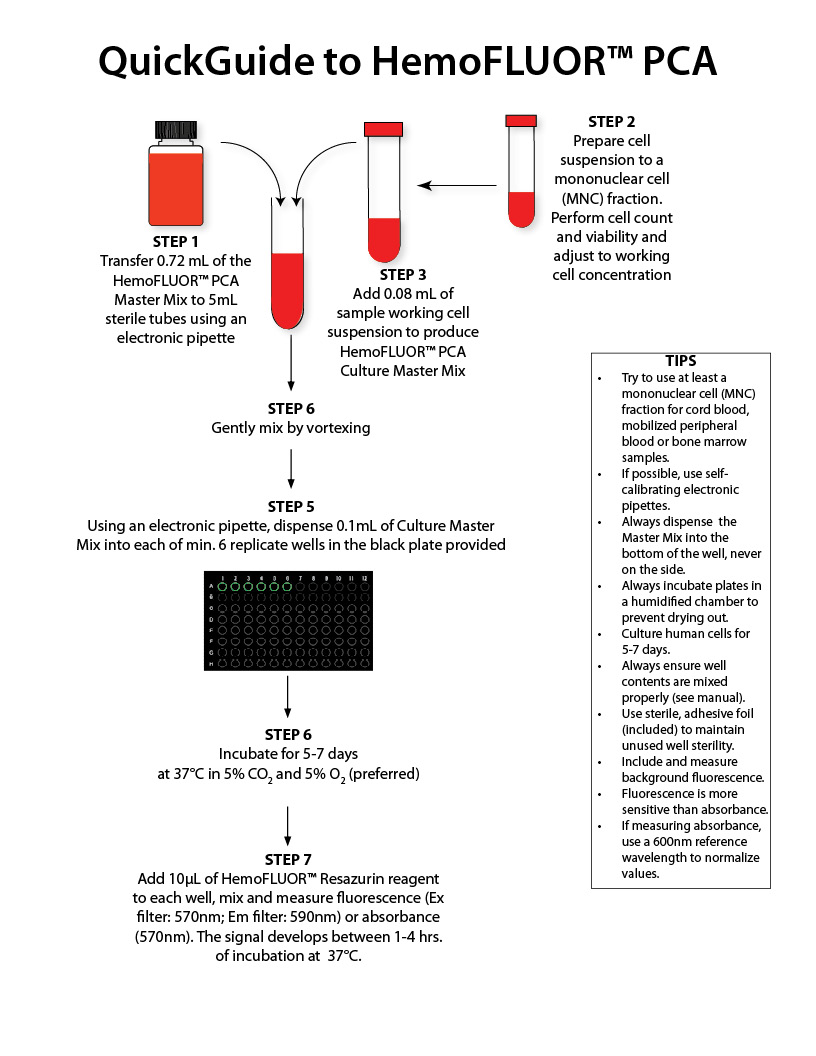
The table below shows the HemoFLUOR™ PCA assays that can replace any methlcellulose, colony-forming unit (CFU) reagent from any competitor. PLEASE NOTE that HemoFLUOR™ PCA incorporates Suspension Expansion Culture™ (SEC™) Technology to replace dispensing error-prone methylcellulose with reagents that use normal pipettes.
| HemoFLUOR™ PCA Number | HemoFLUOR™ PCA Cell Population |
Equivalent CFU Cell Population | Equivalent MethoCult™ Reagent |
Growth Factor Cocktail |
|---|---|---|---|---|
| PCA1 | SC-GEM 3 | CFC-GEM 3 | H4434 "Classic" | EPO, GM-CSF, IL-3, SCF |
| PCA2 | SC-GEM 2 | CFC-GEM 2 | H4034 "Optimum" | EPO, GM-CSF, G-CSF, IL-3, SCF |
| PCA3 | P-GM 1 | GM-CFC 1 | H4534 "Classic" | GM-CSF, IL-3, SCF |
| PCA4 | P-GM 2 | GM-CFC 2 | H4035 "Optimum" | GM-CSF, G-CSF, IL-3, SCF |
| PCA5 | SC-GEMM 3 | CFC-GEMM 3 | H4435 "Enriched" | EPO, GM-CSF, G-CSF, IL-3, IL-6, SCF TPO(#) |
| PCA6 | SC-GEMM | CFC-GEMM | Methocult "Express" | Recombinant cytokines EPO |
(#) denotes that the Preferred Cell Systems™ formulation of this product includes TPO that stimulates the production of megakaryopoiesis. MethoCult™ and other competitor formulations of this specific product do not include TPO, do not stimulate the production of megakaryocytes and therefore do not detect the CFC-GEMM stem cell population.
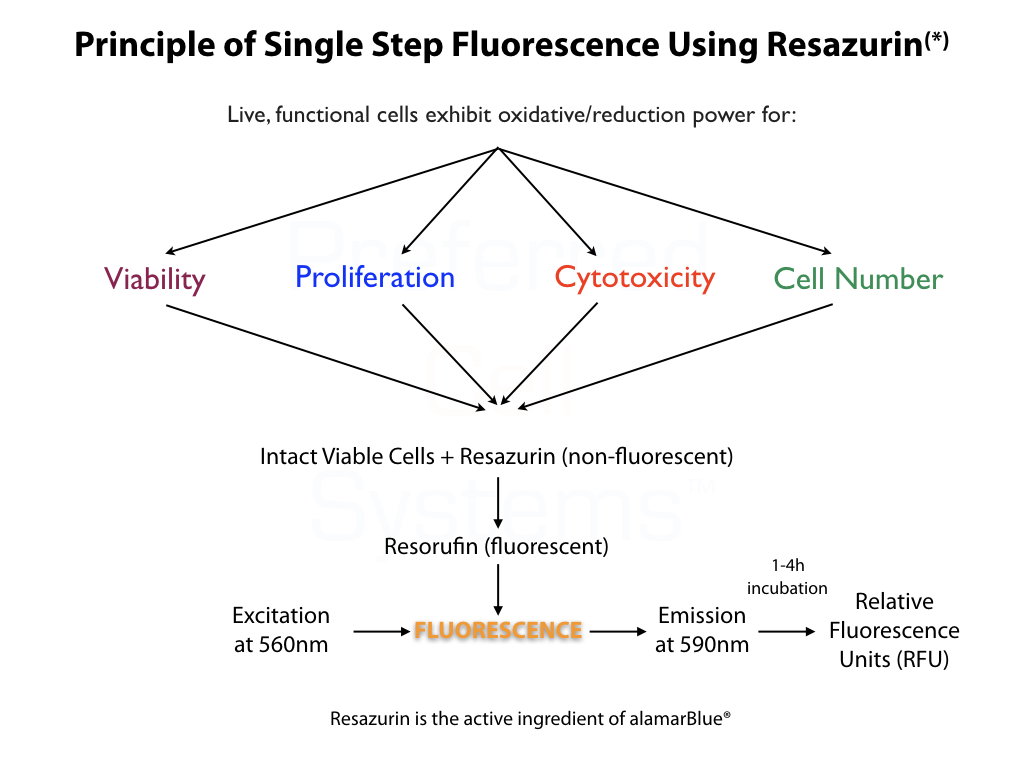
- Incorporates the measurement of non-fluorescent Resazurin (alimaBlue™) to fluorescent resorufin in an oxidation/reduction reaction that occurs only in live cells. The amount of fluorescence (relative fluorescence units, RFU) is dependent on the metabolic status of the cells and therefore is not only a measure of metabolic viability, but cell proliferation or inhibition.
- After culture, add 10μL of reagent, mix and measure signal between 30 minutes and 4 hours in a fluorescence plate reader.
- Uses a plate fluorometer or multimode plate reader to measure fluorescence with an excitation filter 560nm and an emission filter of 590nm.
- Mobilized peripheral blood
- Umbilical cord blood
- Bone marrow
- Purified cell populations for any of the above tissues
- Standardization of the CFU-GM assay using hematopoietic growth factors. J. Hematotherapy:6:191-192 (1997)
- Development of a novel assay to evaluate the functional potential of umbilical cord blood progenitors. Transfusion. 48:620-628 (2008).
- Potency, Proliferation and Engraftment Potential of Stem Cell Therapeutics: The Relationship between Potency and Clinical Outcome for Hematopoietic Stem Cell Products. J Cell Sci Therapy. (2013).
- Detecting primitive hematopoietic stem cells in total nucleated and mononuclear cell fractions from umbilical cord blood segments and units. J Translational Medicine 13:94 (2015)
- Improving quality and potency testing for umbilical cord blood: A New Perspective. Stem Cells Translational Medicine. 4:967-973 (2015)
- Hematopoietic stem cell potency for cellular therapeutic transplantation. In: Hematopoietic Stem Cells, Ed. Rosana P. Camacho. Published by: InTech Open Access Publisher. ISBN 978-953-307-746-8. (2011).
- Measuring the Potency of a Stem Cell Therapeutic. In: Stem Cell Protocols. Methods in Molecular Biology, 1235, Ed. Rich IN. Published by Human Press (2015).
- Bioluminescence Potency Measurement of Cellular Therapy Products. In: Cellular Therapy: Principles, Methods, and Regulations, 2nd Edition, Eds. Areman EM and Loper K. Published by AABB.
- Present Cord Blood Testing Fails to Determine if the Stem Cells Used for Transplantation are of High Quality and PotencyThe Parents Guide to Cord Blood Foundation
- The Difference Between Stem Cell Viability and Potency: A Short Guide for Parent and Patients. The Parents Guide to Cord Blood Foundation
For Research Use Only. Not for clinical diagnostic use.
A fluorescence plate reader or multiparameter plate reader with an excitation filter at 560nm and an emission filter at 590nm.
- HemoFLUOR™ PCA Master Mix
- Resazurin reagent
- 3% SDS solution to stop the reaction
- Resorufin to standardize the assay
- Non-sterile, 96-well plate for assay standardization
- Sterile, 96-well plate(s)
- Sterile, adhesive foil covers so that unused wells remain sterile