STEMpredict™
A Standardized and Validated, 3-Day Hematopoietic Stem Cell Assay to Triage High from Low Quality Cell Therapy Products Prior to Cryopreservation
Buy STEMpredict™ Assay Kits
STEMpredict™
A 3-Day HALO® Style Assay to Triage High from Low Quality Human Hematopoietic Cells Prior to Cryopreservation
| Cell Population | Tissue | Formulation | Catalog Number | Quantity | |
|---|---|---|---|---|---|
| Stem Cells | |||||
| Background + Stem Cells | Any. Low-throughput | Low serum | K2-SP-1 | 1 Kit | |
| Background + Stem Cells | Any. High-throughput | Low serum | K3-SP-1 | 1 Kit | |
| Stem Cells | |||||
| Background + Stem Cells | Any. Low-throughput | Serum-Free | K2SF-SP-1 | 1 Kit | |
| Background + Stem Cells | Any. High-throughput | Serum-Free | K3SF-SP-1 | 1 Kit | |
Download the Stem Cell Product Quality Assay Flyer
STEMpredict™ - Uses, Benefits and Characteristics

- Separate high from low quality stem cell therapeutic products in just 3 days.
- For umbilical cord blood and mobilized peripheral blood. Can also be used for bone marrow.
- Fresh or frozen samples(1).
- Accurate, single dose, cell proliferation ratio data predicts high from low growth-potential units.
- Fully standardized and validated ATP bioluminescence readout according to FDA guidelines.
- Profieciency testing performed during the assay standardization procedure. No addition proficiency testing required.
- Measurement assurance parameters provide proficiency and the trust in your results.
- Makes cell quality using TNC, viability, CD34 or CFU assays obsolete.
- Suspension Expansion Culture™ (SEC™ Technology allows automated use of 96-well plates or 384-well plates for true high-throughput capability for large numbers of samples.
- SEC™ Technology means more accurate dispensing, smaller sample and reagents volumes with faster (automated) setup.
- Most rapid stem cell functional assay available.
- Let the plate reader measure and calculate results in 5 minutes or less.
- Everything is included in the assay kit to culture and measure stem cell quality. Just prepare and add cell sample.
- Always reliable and reproducible results.
- Easy to use, time efficient and cost-effective.
- Easy to learn in just 1 day.
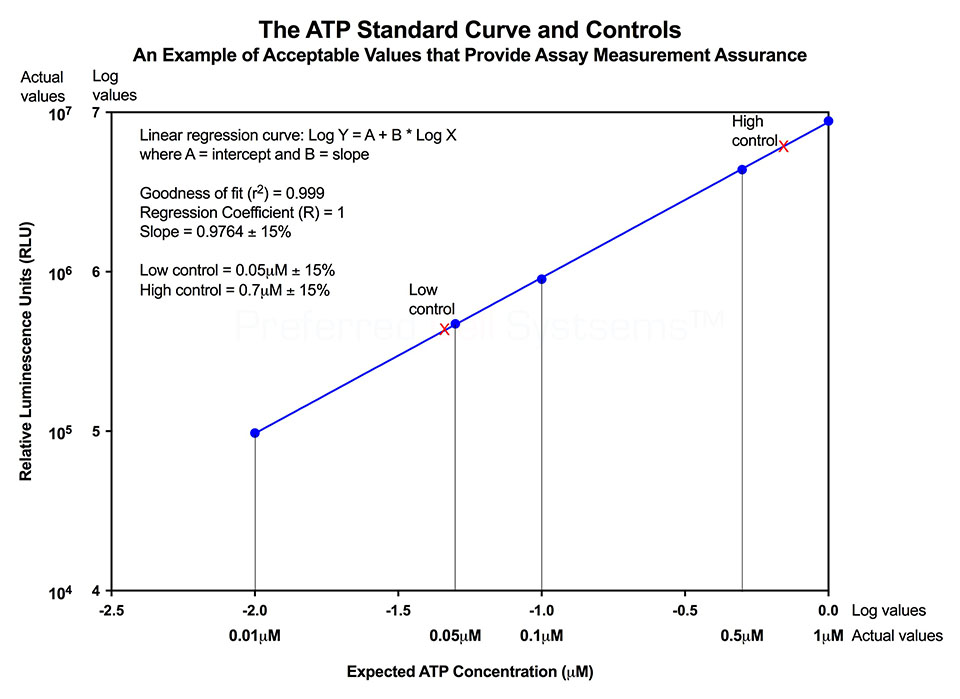
No additional proficiency testing is required if the calibration and standardization procedure is performed. The values you obtain from each calibration and standardization can be logged and used for certification that the assay has been performed correctly and that the results are trustworthy.
- Umbilical cord blood
- Mobilized peripheral blood
- Bone marrow
- Purified cells from any of the above
The recommended cell purity is a mononuclear cell (MNC) fraction or higher purity. A total nucleated cell (TNC) fraction is not recommended as this contains high concentrations of cell impurities, such as red blood cells, neutrophils, platelets and other cells that dilute, mask and severely underestimate and even inhibit the detection of rare primitive stem cells.
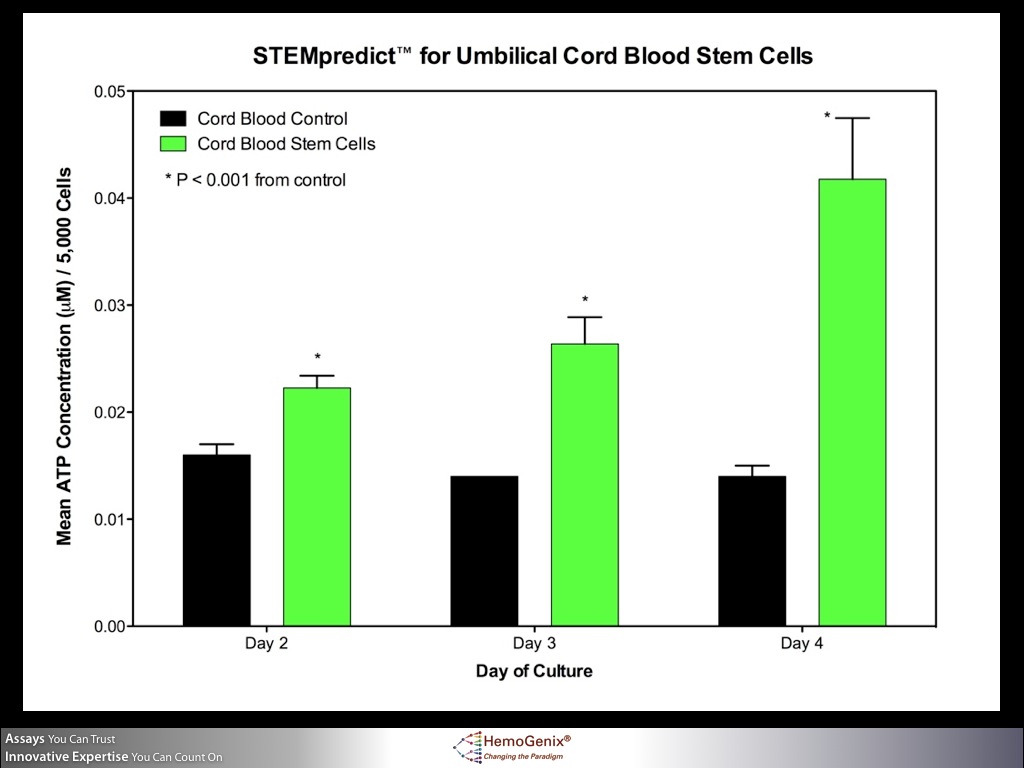
STEMpredict™ for Cord Blood Stem Cells
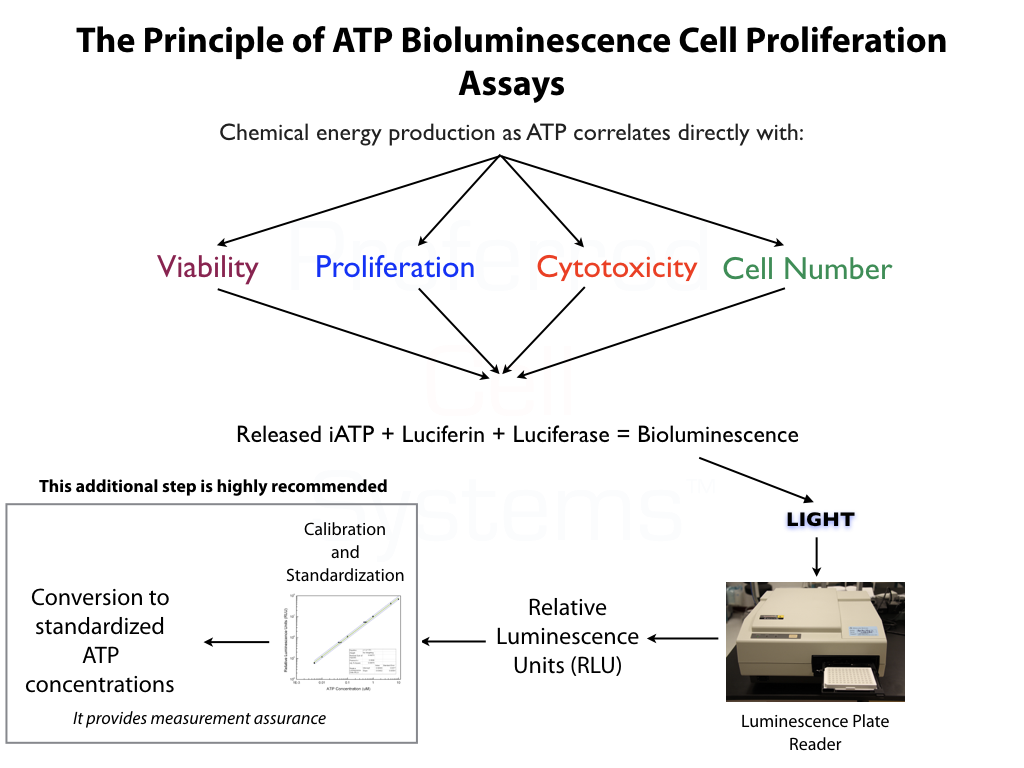
The graph shows an example of how STEMpredict™ predicts stem cell growth, functionality and viability for umbilical cord blood cells. If cells are metabolically viable (which is not the same as being viable by dye exclusion methods), they will demonstrate cellular and mitochondrial integrity by producing intracellular ATP (iATP). The iATP concentration, in turn, directly correlates with proliferation ability. If a quantitative difference in proliferation ability between stimulated and unstimulated (background) stem cells exists, as shown in the graph, the stem cells from the product are predicted to proliferate and grow. In this way, STEMpredict™ not only detects whether the cells are metabolically viable, but also whether they will exhibit cellular functionality and proliferate. The assay therefore measures three parameters simultaneously.
This same concept and principle is applied to mobilized peripheral blood, bone marrow and purified hematopoietic cell products. It is also the cornerstone of all Preferred Cell Systems™ cellular therapeutic assays.
For Research Use Only. Not for clinical diagnostic use.
Luminescence or multimode plate reader with "glow" luminescence measuring capability.
- STEMpredict™ stem cell and control Master Mix
- ATP standard
- ATP high and low controls
- ATP Enumeration Reagent
- Sterile, 96- or 384-well plates for cell culture
- Non-sterile 96-well plates (for assay calibration and standardization)
- Sterile, adhesive foil covers to maintain sterility in unused wells
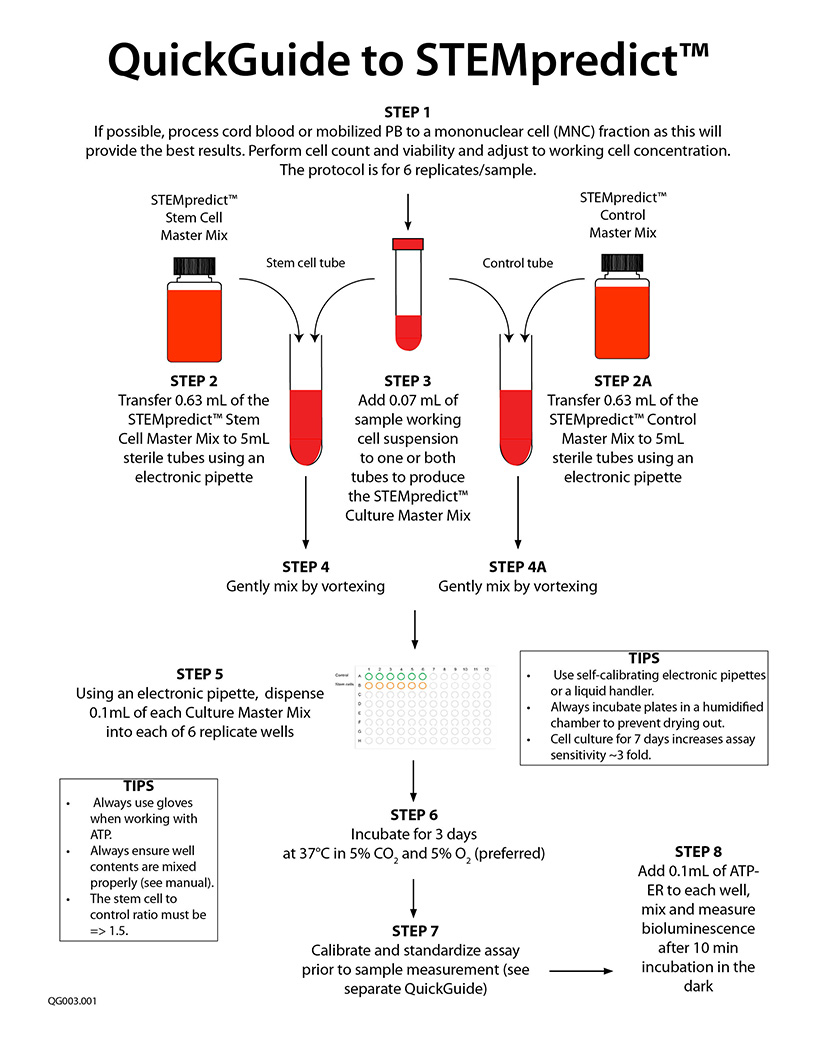
A specific video tutorial on using the STEMpredict™ is not yet available. Please scroll down and check out the QuickGuide and Technical Manual (if available). Below, are the links to perform the ATP bioluminescence readout:
How to Calibrate and Standardize an ATP Bioluminescence Assay - Part 1
How to Calibrate and Standardize an ATP Bioluminescence Assay - Part 2
Proficiency Testing of Hematopoietic Cellular Therapy Products
Download the ATP Optimization Kit Protocol for First-Time Users
Download the Instrument Setup and RLU to ATP Conversion Procedure Download Certificate of Analysis (CoAs) for ATP Enumeration Reagent (ATP-ER)
Download Certificate of Analysis for ATP Stanadrds
Download Certificate of Analysis for ATP Controls
Download Certificate of Analysis of ATP Reconstitution Reagent
Download Certificate of Analysis for Sterile 96-Well Plates
Download Certificate of Analysis for Non-Sterile, 96-Well Plates
Download Certificate of Analysis for Sealing Films
Download Certificate of Analysis for IMDM
When ordering STEMpredict™, please copy the Catalog Number, click on Catalog Number Search and paste the number, followed by Search. You will be taken directly to the desired product.
| Plate Format | Catalog Number | Format | Number of Plates/Kit |
|---|---|---|---|
| 96-well plate. 0.1mL/well assay | K2-SP-1 | Low serum | 1 |
| 96-well plate. 0.1mL/well assay | K2SF-SP-1 | Serum-Free | 1 |
| 384-well plate. 0.025mL/well assay | K3-SP-1 | Low serum | 1 |
| 384-well plate. 0.025mL/well assay | K3SF-SP-1 | Serum-Free | 1 |
To perform STEMpredict™, high throughput, a liquid handler (robot) is required
For bulk orders, please contact Preferred Cell Systems